Author/s: V. Terrasson, J. G. Planas, D. Prim, F. Teixidor, C. Viñas, M. E. Light and M. B. Hursthouse
Reference: Chemistry – A European Journal, 2009, 15, 12030-12042.
Abstract:
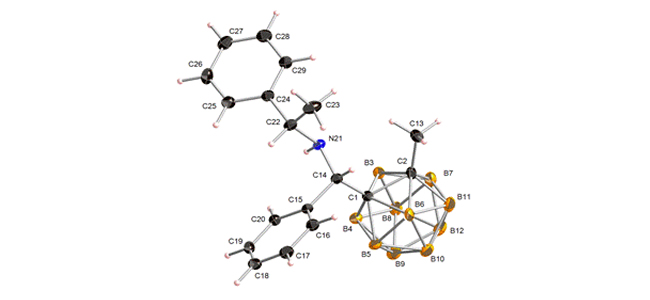
The synthesis of new aminobenzyl-o-carboranes in a practical three-step procedure is described. The strategy involved the preparation of o-carboranylmethanols followed by chlorination and nucleophilic amination. The expected amines were obtained in good overall yields. The synthetic pathway was also extended to the synthesis of new enantiopure carborane-amine combinations.
